Background
Chalk used in school classrooms comes in slender sticks approximately .35 of an inch (nine millimeters) in diameter and 3.15 inches (80 millimeters) long. Lessons are often presented to entire classes on chalkboards (or blackboards, as they were originally called) using sticks of chalk because this method has proven cheap and easy.
As found in nature, chalk has been used for drawing since prehistoric times, when, according to archaeologists, it helped to create some of the earliest cave drawings. Later, artists of different countries and styles used chalk mainly for sketches, and some such drawings, protected with shellac or a similar substance, have survived. Chalk was first formed into sticks for the convenience of artists. The method was to grind natural chalk to a fine powder, then add water, clay as a binder, and various dry colors. The resultant putty was then rolled into cylinders and dried. Although impurities produce natural chalk in many colors, when artists made their own chalk they usually added pigments to render these colors more vivid. Carbon, for example, was used to enhance black, and ferric oxide (FeaCh) created a more vivid red.
Chalk did not become standard in schoolrooms until the nineteenth century, when class sizes began to increase and teachers needed a convenient way of conveying information to many students at one time. Not only did instructors use large blackboards, but students also worked with individual chalkboards, complete with chalk sticks and a sponge or cloth to use as an eraser. These small chalkboards were used for practice, especially among the younger students. Pens dipped in ink wells were the preferred tool for writing final copy, but these were reserved for older students who could be trusted not to make a mess: paper—made solely from rags at this time—was expensive.
An important change in the nature of classroom chalk paralleled a change in chalkboards. Blackboards used to be black, because they were made from true slate. While some experts advocated a change to yellow chalkboards and dark blue or purple chalk to simulate writing on paper, when manufacturers began to fashion chalkboards from synthetic materials during the twentieth century, they chose the color green, arguing that it was easier on the eyes. Yellow became the preferred color for chalk.
Almost all chalk produced today is dustless. Earlier, softer chalk tended to produce a cloud of dust that some feared might contribute to respiratory problems. Dustless chalk still produces dust; it’s just that the dust settles faster. Manufacturers accomplish this by baking their chalk longer to harden it more. Another method, used by a French company, is to dip eighty percent of each dustless chalk stick in shellac to prevent the chalk from rubbing off onto the hands.
Raw Materials
The main component of chalk is calcium carbonate (CaCCh), a form of limestone. Limestone deposits develop as coccoliths (minute calcareous plates created by the decomposition of plankton skeletons) accumulate, forming sedimentary layers. Plankton, a tiny marine organism, concentrates the calcium found naturally in seawater from .04 percent to 40 percent, which is then precipitated when the plankton dies. The base of pastel chalks is calcium sulfate (CaSOi), which is derived from gypsum (CaS04»2H20), an evaporite mineral formed by the deposition of ocean brine; it also occurs disseminated in limestone. Chalk and dehydrated gypsum thus have similar origins and properties. Pastels also contain clays and oils for binding, and strong pigments. This mixture produces sticks that write smoothly without smearing and draw better on paper than on chalkboards. Although great care is taken to eliminate contaminants when chalk is manufactured, some impurities inherent to the mineral remain. Chief among these are silica, alumina, iron, phosphorus, and sulfur. In less significant, amounts, manganese, copper, titanium, sodium oxide, potassium oxide, fluorine, arsenic, and strontium may also occur.
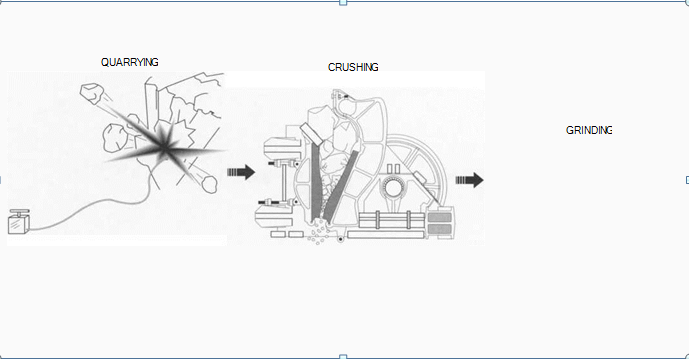
(To make chalk, limestone is first quarried, generally by an open pit quarry method. Next, the limestone must be crushed. Primary crushing, such as in a ¡aw crusher, breaks down large boulders; secondary crushing pulverizes smaller chunks into pebbles. The limestone is then wet-milled with water in a ball mill— a rotating steel drum with steel balls inside to further pulverize the chalk. This step washes away impurities and leaves a fine powder)
The Manufacturing Process
Quarrying limestone
1 Approximately 95 percent of the lime-stone produced in the United States is quarried. After a sufficient reserve (twenty-five years’ worth is recommended) has been prospected, the land that covers the deposit is removed with bulldozers and scrapers. If the chalk is close to the surface, an open shelf quarry method can be used; however, this is very rare. Usually an open pit quarry method is used instead. In this method, holes are drilled into the rock, explosives are placed inside, and the rock is blown apart. Depending on the nature of the deposit, a pit can be enlarged laterally or vertically.
Pulverizing the chalk
2 Once comparatively large chunks of limestone have been quarried, they need to be transported to crushing machines, where they are pulverized to meet the demands of the chalk industry. The first step is primary crushing. Various crushers exist, but the principle is the same: all compress the stone with jaws or a cone, or shatter it through impact. Secondary crushing is accomplished by smaller crushers that work at higher speeds, producing pebbles which are then ground and pulverized.
3 The next phase, wet grinding, washes away impurities. It is used to make the fine grade of limestone necessary to make chalk suitable for writing purposes. Wet grinding is carried out in ball mills—rotating steel drums with steel balls inside that pulverize the chalk until it is very fine.
(After grinding, the chalk particles are sifled over vibrating screens to separate the finer particles. The particles are then mixed with water, extruded through a die of the proper size, and cut to the proper length. Finally, the chalk is cured in an oven for four days)
Dehydrating gypsum
4 Gypsum, like limestone, is also quarried and pulverized. The major difference in processing gypsum is that it must be dehydrated to form calcium sulfate, the major component of colored chalk. This is done in a kettle, a large combustion chamber in which the gypsum is heated to between 244 and 253 degrees Fahrenheit (116-121 degrees Celsius). It is allowed to boil until it has been reduced by twelve to fifteen per-cent, at which point its water content will have been reduced from 20.9 percent to between 5 and 6 percent. To further reduce the water, the gypsum is reheated to about 402 degrees Fahrenheit (204 degrees Celsius), at which point it is removed from the kettle. By now, almost all of the water has evaporated, leaving calcium sulfate.
Sifting, cleaning, and shipping the chalk
5 The particles of chalk or calcium sulfate are now conveyed to vibrating screens that sift out the finer material. The ensuing fine chalk is then washed, dried, packed in bags, and shipped to the manufacturer. Upon receiving chalk or calcium sulfate, the chalk factory usually grinds the materials again to render them smooth and uniformly fine.
Making white classroom chalk
6 To make white classroom chalk, the manufacturer adds water to form a thick slurry with the consistency of clay. The slurry is then placed into and extruded from a die—an orifice of the desired long, thin shape. Cut into lengths of approximately 24.43 inches (62 centimeters), the sticks are next placed on a sheet that contains places for five such sticks. The sheet is then placed in an oven, where the chalk cures for four days at 188 degrees Fahrenheit (85 degrees Celsius). After it has cured, the sticks are cut into 80 millimeters lengths.
Making colored classroom chalk
7 Pigments (dry, natural, colored materials) are mixed in with the calcium carbonate while both are dry (the procedure is similar to sifting flour and baking powder together before adding liquid, as in a cake recipe). Water is then added to the mixture, which is then baked in the same manner as white classroom chalk.
Making pastels
8 Another manufacturing method is used for pastels, the chalks used for art drawing. The procedure resembles that used for colored classroom chalk, but calcium sulfate. Chalk is used instead of calcium carbonate. In addition, the dry material is mixed with clay and oils, and more pigments are added to produce a slurry that has the consistency of toothpaste. Because the final products must be relatively moist, pastels are usually air- dried rather than baked.
Boxing the chalk
9 Placed in small boxes, the completed chalk sticks are stacked in large boxes to be shipped to supply stores.
Quality Control
Chalk that is intended for the classroom must undergo stringent tests in order to perform well and be labeled nontoxic. All incoming materials are tested for purity before being used. After the chalk has been made into sticks, one stick from each batch is selected for tests. The density and break strength of the sample stick are determined. The sample is then used to write with, and the quality of the mark is studied. Erasability is also studied. First, the chalk mark is erased using a dry eraser, and the quality of erasure is examined. Then, the chalkboard is washed, and again the amount of chalk left on the board is examined. Furthermore, a sample from each batch is kept for five years so that it can be inspected if in the future its quality is questioned.
Chalk for classroom use adheres to the American National Standards Institute performance standards. Written specifications state the proper length of the chalk stick, as well as how many sticks should go in a box. On November 18, 1990, a Federal Act (Public Law 100-695) went into effect, mandating that all art materials sold in the United States must be evaluated by a qualified toxicologist who must then issue a label explaining their toxicity. Toxicologists are concerned not with cost but with safety, and they must consider many factors before granting approval. Each ingredient, the quantity in which it is used, and its possible adverse reactions with other ingredients are studied. The product’s size and packaging, its potential harm to humans, and its tendency to produce allergic reactions are also considered. Toxicologists also take into account the products use and potential misuse, as well as all federal and state regulations. Formulas for every color and every formula change must meet approval.
Classroom chalk is labeled “CP [certified product] nontoxic” if it meets the standards of the Art and Craft Materials Institute, a nonprofit manufacturers’ association. This label certifies that art materials for children are nontoxic and meet voluntary standards of quality and performance, and that the toxicity of art materials for adults has been correctly labeled. The CP seal also indicates that the product meets standards of material, workmanship, working qualities, and color developed by the Art and Craft Materials Institute and others such as the American National Standards Institute and the American Society for Testing and Materials (ASTM). To ensure honesty, most chalk manufacturers are tested at random by an independent toxicologist, who checks to see that they are meeting nontoxic standards. Most manufacturers conform to such exacting standards because knowledgeable schools will not purchase chalk that is not properly labeled.
The Future
Many people consider using chalk and chalk-boards to present material outdated. Some experts claim that teachers have stubbornly resisted new technologies that could improve teaching—and eliminate the chalkboard entirely. A study which recently investigated whether teaching with overhead projectors was more effective than using chalkboards concluded that chalkboards were more interactive, progressive, and fruitful.
A development much in the educational news lately is the electronic chalkboard. In place of a regular chalkboard, a teacher uses a large TV screen, inputting materials from a computer terminal. In a more advanced scenario, each student uses a terminal, to which the teacher sends information from a master computer. Experts claim that such set-ups are more visually exciting to students, more versatile than the old-fashioned chalkboards, cleaner than dusty chalk, easier for the teacher to use, and better able to present more complex material through the use of graphics and animation. Many studies on the feasibility of electronic chalkboards have been made, however, and most seem to favor keeping the traditional chalkboard, at least for now. Electronic chalkboards that are sophisticated and easily readable lie beyond the budget constraints and technological capabilities of most schools. Further, studies of the electronic system’s effectiveness report that teachers who use it spent more time preparing their lessons, teachers and students were less interactive, students were dissatisfied with the electronic chalkboards, and the new devices divided the students’ attention between the screen and the teacher conveying the information. Although the enthusiasm for electronic blackboards in some areas remains high, chalk use in the classroom is guaranteed for some time to come.,
Periodicals
Toth, Beth. “Jeanne Otis: A Color Dialogue,” Ceramics Monthly. January, 1988, p. 40. —Rose Secrest
Where To Learn More
Books
Boynton, Robert. Chemistry and Technology of Lime and Limestone. John Wiley & Sons, 1980.